Hoe wij klinisch onderzoek doen
How we conduct clinical research
Without clinical research, we cannot provide a scientifically based answer with regard to the safety and efficacy of the innovative treatments that we have developed during the R&D process. At Janssen, we conduct this clinical research in accordance with the highest quality standards. Volunteers and patients are indispensable in this process. By cooperating intensively with doctors, researchers and hospitals in order to carry out the clinical studies and then sharing the results transparently, we aim to bring the research and scientific knowledge to a higher level. In other words, it is an essential step in the process of making our contribution to improving patient care and the healthcare system.
A large part of Janssen's phase 1 research is carried out in the Janssen Clinical Pharmacology Unit (CPU), in the Antwerp Hospital Network Jan Palfijn hospital in Merksem. This research center plays an important role in the development of new treatments, but also in the further development of existing medicines, therapies and vaccines.
Thanks to the rich portfolio of new treatments under development, the presence of the CPU in Merksem and the set-up of a large number of studies in cooperation with hospitals in various disease areas, Janssen fulfils an important role in the clinical research field in Belgium. Belgium is one of the frontrunners in Europe, partly thanks to an ambitious and proactive government in this area and the favorable regulatory framework. We distinguish ourselves from other European countries by our agility and the achievement of particularly competitive timelines, the quality of the data supplied, the possibility of scientific advice in the development of a study protocol and strong scientific expertise. This expertise stems from the unique collaboration between academic research centers, university hospitals and the pharmaceutical industry itself. In addition, research and development (R&D) are high on the agenda of the Belgian government. This shared vision of clinical research and the intensive collaboration between researchers, government and industry has ensured that Belgium has taken a leading role in Europe and even worldwide. In 2020, Belgium confirmed its position as a European leader in the field of clinical trials, with 503 approved applications, 80% of which were initiated by pharmaceutical companies such as Janssen.
Healthy volunteers and patients are indispensable in clinical research. Their safety and well-being are always paramount, and we inform them extensively before the study begins. For example, participants are informed of all study procedures and the potential risks of drug research, and they must give prior written consent to participate in a clinical study. This is done through an information and consent form. In addition, all our clinical studies must comply with internationally accepted ethical and scientific guidelines for Good Clinical Practice (GCP)1, which guarantee the rights, safety, and well-being of the study participants. Afterwards, we share this research data in full transparency at various levels.
"Belgium has a very good reputation in Europe in the field of clinical research, especially thanks to the speed with which we initiate clinical studies. But we are also known for the high quality of our clinical studies and the expertise that doctors have in conducting them."

Kirsten Vandewalle
Clinical R&D Director
Phases of clinical research
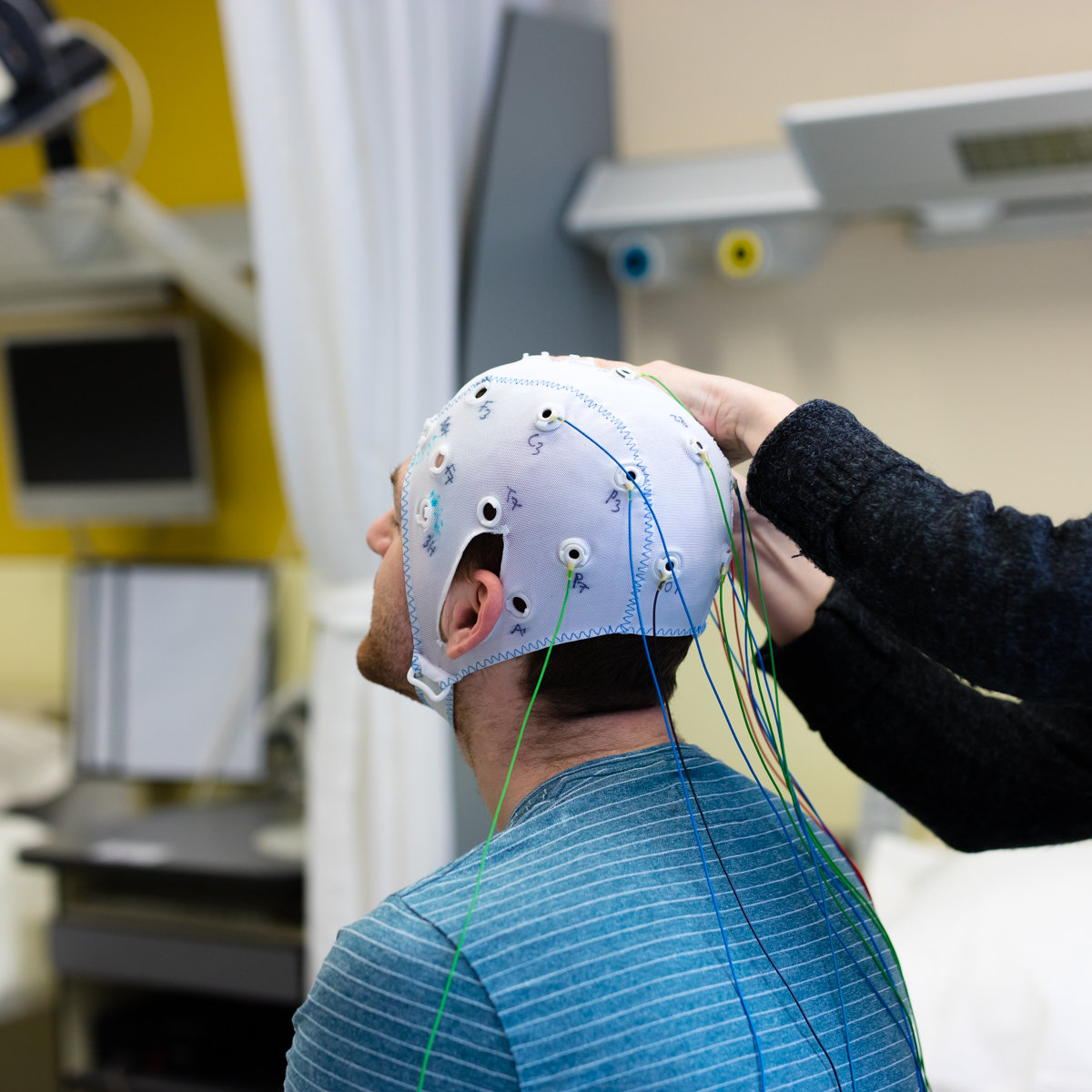
Clinical research consists of several phases. First, the new treatment is tested in small groups of healthy people, except in cancer research, for example. Later, one starts with a larger patient population. Each individual clinical trial must first be evaluated by an ethics committee and the appropriate authorities, who then issue an official approval for the study.
Phase 1 studies focus on safety and are usually conducted on a small number of healthy volunteers or a very limited group of patients with a very specific condition. They last about a year. In vaccine research, the numbers in phase 1 are already somewhat higher. The goal is to find out what our bodies do with the drug or vaccine and what immediate side effects may occur. This phase also takes the first steps toward determining a safe dose of the future, potential treatment.
Phase 2 studies are examining for the first time whether the treatment has a positive therapeutic effect in a limited group of patients with a specific condition. In addition, the dosage is further fine-tuned, and side effects (if any) are further investigated. In vaccine studies, a study is conducted in a larger group of healthy volunteers (up to 5,000 people) to determine which dosage produces the desired immune response.
Phase 3 studies are often international studies in numerous participating centers with several hundred to even thousands of patients or - in the case of vaccines - healthy volunteers. At this stage, the efficacy of the therapy, drug or vaccine is examined and additional data on safety are collected. In this phase the effectiveness of the treatment is compared with either a placebo or with the current standard treatment.
"Janssen wants to offer therapies tailored to the patient. But the data you need for that are scattered across different sources. The application of this data must comply with current privacy legislation, so that the person behind the data cannot be identified. This is crucial to protect the patient."

Brahem El Haddioui
Senior Manager Benelux Global Privacy

David Fauvart
Director Clinical Innovation
6.5
On average, the development of an innovative treatment - from lab to patient - takes about 12 to 13 years. After the preclinical phase, the various clinical research phases begin. Together, these take approximately 6.5 years.
37
In 2020, Janssen has more than doubled the number of participants in our clinical development programs. For example, 37 new study protocols were set up at 72 different sites in Belgium (out of a total of 237 active studies). This led to 1,680 new participants out of a total of 2,565. The accelerated research on our vaccine is the main explanation for this.
Collaboration in clinical research

As in the other phases of our drug development, collaboration is central to clinical research. Pharmaceutical companies, researchers in hospitals, healthcare providers and participants work together to gather knowledge about the safety and effectiveness of a future treatment.
Pharmaceutical companies
Pharmaceutical companies develop a drug candidate, in some cases together with researchers in universities. For example, they first go through all the phases of preclinical research, which you could read about in Chapter 2. When a new treatment is ready for clinical studies, a study protocol is written on the basis of a research question, with a detailed description of all the procedures to be carried out. An information and consent form is prepared for study participants, describing all procedures, risks, participant rights, and the course of the study. The study protocol, together with all available safety information and patient information, is submitted to the authorities and an ethics committee, who must give their approval before the study can begin. Once the study is approved, the physician investigators are trained on the study. The study is further followed up by a representative of the "sponsor" - in this case Janssen - in order to ensure that the correct procedures are followed and that the rights and welfare of the participants can be guaranteed.

COVID-19: Development of our corona vaccine

COVID-19: Development of our corona vaccine
Belgium was involved in the clinical research for the candidate vaccine against COVID-19 from the very beginning.
From the moment that the genetic code of the coronavirus became known in January 2020, Janssen researchers have worked tirelessly to develop a vaccine against COVID-19. The development and research phases were completed rapidly without compromising our quality standards. The various phases of clinical trials were initiated in parallel, but not before the safety of the vaccine had been demonstrated in the important phase 1 study.
After our most promising vaccine candidate was selected in the preclinical development phase in late March 2020, it was administered for the first time in healthy volunteers in the United States and... in Belgium in July 2020. This marked the beginning of phase 1/2a clinical trials, which examined the safety profile and immune response of the vaccine candidate in a limited group of volunteers. Following the positive interim results of this phase, which demonstrated the safety of the vaccine, we began the phase 2 and phase 3 studies in the second half of September 2020 to demonstrate the efficacy of the vaccine in a broader population.
One of our large-scale phase 3 studies 'ENSEMBLE-1' took place on three different continents in more than 44,000 volunteers, 1/3rd of whom were older than 65 years. Thanks to international collaborations with the participating study centers, we were able to investigate the safety and efficacy of the vaccine candidate on this large scale in a short period of time. At the end of January 2021, we published the interim results of our phase 3 study. These results demonstrated the efficacy of our vaccine in reducing both mild and severe disease progression of COVID-19 and demonstrated that it is well tolerated by study participants. Based on these results, we were able to apply for approval of the vaccine with the Food and Drug Administration (FDA) and European Medicines Agency (EMA) within a year of starting preclinical studies. A daring feat that would not have been possible without the boundless commitment of all our employees and the intensive cooperation with the competent authorities. The work is not over yet. We will continue to conduct research in order to follow up the effects of the vaccine in daily practice and thus collect 'real world data'. Because COVID-19 is still a relatively new virus that has an erratic course, numerous (follow-up) studies are currently underway.

e-PIL

e-PIL
The safe and correct use of medicines by patients and healthcare professionals is an important priority for Janssen. The paper leaflet in the box is a source of relevant information but must occasionally be adapted to the most recent insights. This takes time, which means that patients - or, in the case of vaccine studies, healthy individuals - do not always immediately have the most recent information available. For this reason, Janssen is working closely with the Federal Agency for Medicines and Health Products (FAMHP) on e-PIL, a digital translation where both the patient and the health care provider can always access the most recent, up-to-date version of the leaflet.
With participants
Our clinical trials are designed to make a difference for patients and society as a whole. Therefore, we include the perspective of patients as much as possible in the design of our clinical trials. For example, we work with a group of patients to provide input on the feasibility of a study protocol, the readability of an information and consent form, or other patient materials. To ensure the best possible feasibility, digital questionnaires, assessments, and wearables are currently increasingly being used where participants can provide input on how they feel in various areas and what side effects, if any, they are experiencing. In the context of "care in the home setting," we are increasingly seeing someone from the research team visit the patient at home to perform certain study-specific actions, such as a blood draw or administering the medication.
"Over the past year, we have further developed and improved our privacy policy. For example, we created an electronic document for informed consent for patients and volunteers participating in a clinical trial. This so-called 'e-consent' is very user-friendly: by using videos and icons, we can now make information much more understandable than purely written text. That is much clearer for the participants"

Brahem El Haddioui
Senior Manager Benelux Global Privacy
Transparency on Clinical Trials
With researchers
With clinical research, we build our knowledge about a new therapy, drug, or vaccine. By sharing the results, we ensure that other researchers can also learn from our work. This helps advance science and thus contributes to overall public health: it contributes to a better understanding of diseases and increases the knowledge needed to develop new, innovative treatments.
We share this research data at various levels. For example, we publish data on our Belgian studies in the publicly available clinical trials database of the Federal Agency for Medicines and Health Products (FAMHP), but also, for example, in scientific journals. In addition, we also publish the information on our studies in the European Register for Clinical Trials (ECTR) 2Yale Open Data Access (YODA) project.
''In my work, I can bring scientific innovation and at the same time be involved with people. Thanks to the innovative nature of our work, we can help patients for whom there are no treatment options at present. The fact that I can contribute to this is something I find extremely beautiful.”

Kirsten Vandewalle
Clinical R&D Director
